 
For example, thallium (Tl) in sedimentary rocks has a different isotopic composition than in igneous rocks and volcanic gases. For helium, A r, abridged°(He) = 4.0026.įor fourteen elements the samples diverge on this value, because their sample sources have had a different decay history. IUPAC also publishes abridged values, rounded to five significant figures. The "(2)" indicates the uncertainty in the last digit shown, to read 4.002 602 ☐.000 002. Typically, such a value is, for example helium: A r°(He) = 4.002 602(2). Of the 118 known chemical elements, 80 have stable isotopes and 84 have this Earth-environment based value. This range is the rationale for the interval notation given for some standard atomic weight values. Standard atomic weight averages such values to the range of atomic weights that a chemist might expect to derive from many random samples from Earth. Non-standardized atomic weights of an element are specific to sources and samples, such as the atomic weight of carbon in a particular bone from a particular archeological site. 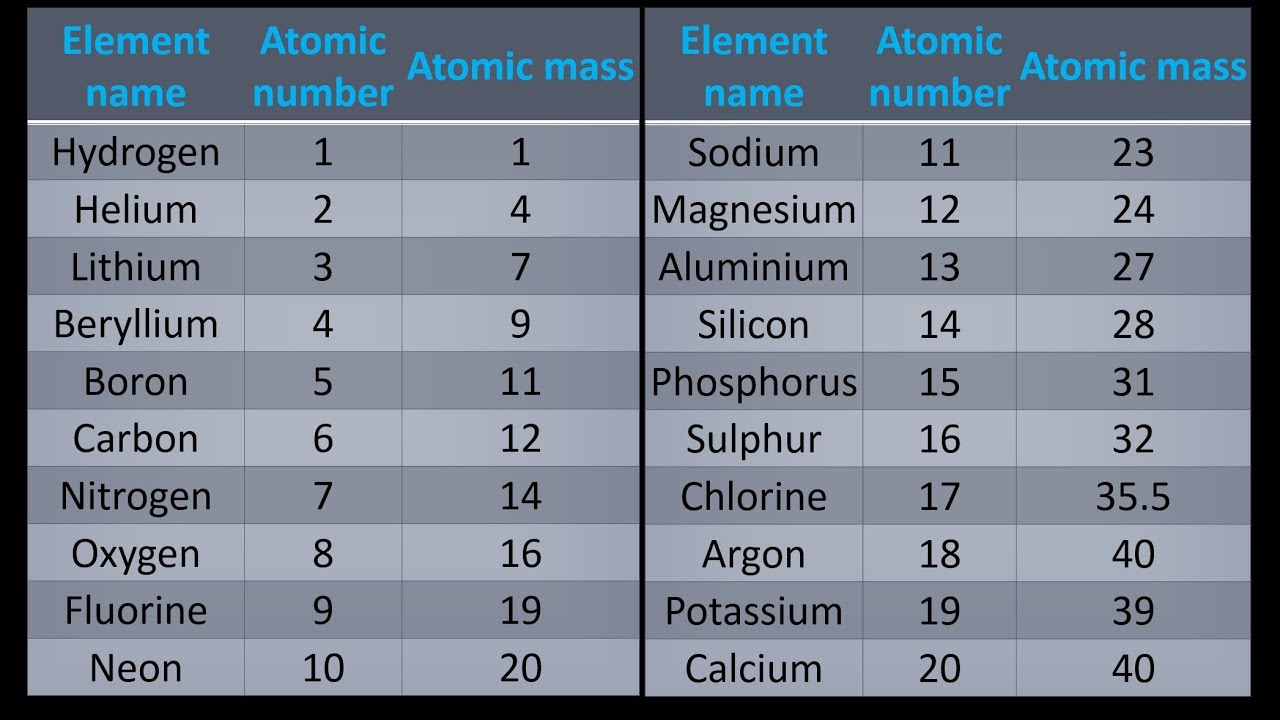
The definition specifies the use of samples from many representative sources from the Earth, so that the value can widely be used as "the" atomic weight for substances as they are encountered in reality-for example, in pharmaceuticals and scientific research. The standard atomic weight of each chemical element is determined and published by the Commission on Isotopic Abundances and Atomic Weights (CIAAW) of the International Union of Pure and Applied Chemistry (IUPAC) based on natural, stable, terrestrial sources of the element. It can be converted into a measure of mass (with dimension M) by multiplying it with the dalton, also known as the atomic mass constant.Īmong various variants of the notion of atomic weight ( A r, also known as relative atomic mass) used by scientists, the standard atomic weight ( A r°) is the most common and practical. īecause relative isotopic masses are dimensionless quantities, this weighted mean is also dimensionless. The standard atomic weight of a chemical element (symbol A r°(E) for element "E") is the weighted arithmetic mean of the relative isotopic masses of all isotopes of that element weighted by each isotope's abundance on Earth. The standard atomic weight ( A r°(Cu)) for copper is the average, weighted by their natural abundance, and then divided by the atomic mass constant m u. The details all of the elements in the Periodic table, the numbers of protons, the numbers of neutrons and the mass numbers of atoms which relate to the elements in the Periodic Table.Not to be confused with Atomic mass. Neon has the Atomic Mass weight of 20.18. The number of protons in atom of neon is therefore 10. Example 3 - mass number of Neon: The element Neon (Symbol Ne) has the Atomic Number of 10.The mass number of silver is therefore 108. Silver has the Atomic Mass weight of 107.87. The number of protons in atom of silver is therefore 47. Example 2 - mass number of Silver: The element Silver (Symbol Ag) has the Atomic Number of 47.The mass number of gold is therefore 197. Gold has the Atomic Mass weight of 196.97. The number of protons in atom of gold is therefore 79. Example 1 - mass number of Gold: The element Gold (Symbol Au) has the Atomic Number of 79.The following examples provide details of how to calculate the mass number. The unique chart below has been created by and details all of the elements in the Periodic table, the numbers of protons, the numbers of neutrons and the mass numbers of atoms which relate to the elements. So, if we know the number of protons and neutrons in an atom we can determine the mass number. Mass Numbers - The Mass Numbers of all of the elements 
Number of Neutrons = Mass Number - Atomic Number Mass Numbers Mass Numbers = Atomic Weight of Element, rounded to nearest whole number Subtracting the Atomic number from the Mass Number equals the number of neutrons in the nucleus. Mass numbers equal the total number of heavy, or massive, particles in the nucleus. The chemical properties of an element are determined by its Atomic Number not its Mass Number which is why atomic numbers are shown on the Periodic table whilst Mass Numbers are not. The Periodic Table with Atomic Mass will give you the atomic weight, or atomic mass, of the elements. The mass number is established by rounding the atomic weight to the nearest whole number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
